
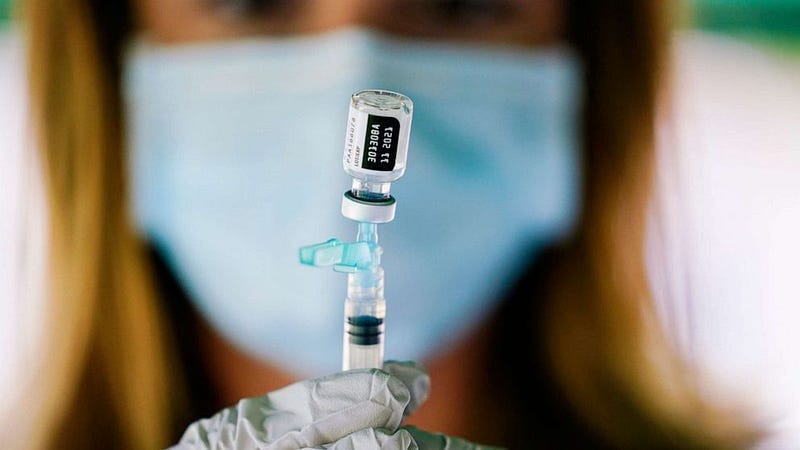
Data shows the Pfizer and BioNTech COVID-19 vaccine is safe and effective for children ages 5 to 11, the companies announced Monday morning.
“We are eager to extend the protection afforded by the vaccine to this younger population, subject to regulatory authorization, especially as we track the spread of the delta variant and the substantial threat it poses to children,” Pfizer chairman and CEO Albert Bourla said in a statement. “Since July, pediatric cases of COVID-19 have risen by about 240 percent in the U.S. — underscoring the public health need for vaccination.”
There were 2,268 participants ages 5 to 11 in the trial, which, while it still followed a two-dose regimen, used a lesser dose than the amount given to people ages 12 and older, for the “safety, tolerability and immunogenicity” of younger children.
“In participants 5 to 11 years of age, the vaccine was safe, well-tolerated and showed robust neutralizing antibody responses,” the companies said in a news release.
Pfizer and BioNTech plan to share their data with the U.S. Food and Drug Administration, the European Medicines Agency and other regulators soon and will submit a request for emergency use authorization in the United States.
“These trial results provide a strong foundation for seeking authorization of our vaccine for children 5 to 11 years old, and we plan to submit them to the FDA and other regulators with urgency,” Bourla said.
Results from two other ongoing trials — one of children ages 2 to 5 and one of children 6 months to 2 years old — are expected as soon as later this year, the companies said.
Pfizer has also received emergency use authorization from the FDA to give a third dose of vaccine to those 12 years of age and older who have been determined to have certain kinds of immunocompromise.
However, a panel advising the federal government on vaccines recently rejected President Joe Biden’s suggestion to roll out booster shots for all Americans, recommending that for now, a third dose should only be given to those 65 and older or those at high risk of severe COVID-19.
In January, Alaska had the highest per capita coronavirus vaccination rate in the nation. Now, hospitals are overwhelmed with COVID-19 patients, and the state’s largest hospital is rationing care.
Vaccine hesitancy and the delta variant have pushed the state’s fragile and limited hospital system to the breaking point.
Providence Alaska Medical Center, the state’s largest hospital, released a letter to the public Tuesday saying that more than 30% of its patients have COVID-19 and the hospital is rationing treatment.
“While we are doing our utmost, we are no longer able to provide the standard of care to each and every patient who needs our help,” wrote Chief of Staff Kristen Solana Walkinshaw on behalf of the hospital’s Medical Executive Committee. “The acuity and number of patients now exceeds our resources and our ability to staff beds with skilled caregivers, like nurses and respiratory therapists.”
Of Alaska’s 120 ICU beds, 106 were filled as of Thursday — leaving only 14 beds available statewide.
Alaska had a strong initial vaccine rollout, delivering doses to remote areas of the state by helicopters, planes, dog sleds and ferries, with additional support from the Indian Health Service and state tribal health system to vaccinate Alaska Natives. Due to the challenges posed by the state’s vast size, it received vaccine allocations monthly as opposed to weekly, giving it the ability to plan ahead and deliver many doses early on.
But, as in the rest of the country, vaccination rates slowly began dropping off over the summer, stagnating with 56.7% of Alaskans fully vaccinated as of Thursday, according to the state’s coronavirus dashboard.
“In terms of why things went stagnant, it does seem like hesitancy is the main factor behind that,” said Jared Kosin, CEO and president of the Alaska State Hospital and Nursing Home Association. “It’s not an access issue. The vaccine’s widely available in Alaska anywhere.”
https://www.periodicoelparamo.com/advert/123movies-watchshang-chi-and-the-legend-of-the-ten-rings-2021hd-full-movie-online-for-free/
https://www.periodicoelparamo.com/advert/123movies-watch-candyman-2021-hd-full-movie-online-for-free/
https://www.periodicoelparamo.com/advert/123movies-watch-free-guy-2021-hd-full-movie-online-free/
https://www.periodicoelparamo.com/advert/123movies-watch-jurassic-hunt-2021-hd-full-movie-online-free/
https://www.periodicoelparamo.com/advert/watchpaw-patrol-the-movie-2021hd-full-movie-online-for-free/
https://www.periodicoelparamo.com/advert/123movies-watch-sweet-girl-2021free-hd-full-movie-online/
https://www.periodicoelparamo.com/advert/123movies-watch-paw-patrol-the-movie-2021-full-hd-movie-online-free/
https://www.periodicoelparamo.com/advert/123movieswatch-firebird-2021full-hd-movie-online-free/
https://www.periodicoelparamo.com/advert/123movies-watch-cinderella-2021-hd-full-movie-online-free/
https://www.periodicoelparamo.com/advert/123movies-watch-after-we-fell-2021-hd-full-movie-online-free/
https://www.periodicoelparamo.com/advert/123movies-watch-belfast-2021-full-movie-online-free/
https://www.periodicoelparamo.com/advert/123movieswatch-kate-2021hd-full-movie-online-free/
https://www.periodicoelparamo.com/advert/watchthe-suicide-squad-2021-hd-full-movie-online-for-free/
https://www.periodicoelparamo.com/advert/123movies-watch-cruella-2021-hd-full-movie-online-free/
https://www.periodicoelparamo.com/advert/123movies-watch-rogue-hostage-2018-hd-full-movie-online-free/
https://www.periodicoelparamo.com/advert/watch-mortal-kombat-legends-battle-of-the-realms-2021full-online-movie-free-hd/
https://www.periodicoelparamo.com/advert/123movies-watch-malignant-2021-hd-full-movie-online-free/
https://www.periodicoelparamo.com/advert/123movies-watch-escape-room-tournament-of-champions-2021-hd-full-movie-online-free/
https://www.periodicoelparamo.com/advert/watchsas-red-notice-2021hd-full-movie-online-for-free-123movies/
https://www.periodicoelparamo.com/advert/watch-the-night-house-2021full-hd-movie-online-free-downloads/
https://www.periodicoelparamo.com/advert/watchafterlife-of-the-party-2021-hd-full-movie-online-for-free-123movies/
https://vocus.cc/article/61487294fd89780001f1448d
https://vocus.cc/article/614872f5fd89780001f146a8
https://vocus.cc/article/614871e2fd89780001f14087
https://vocus.cc/article/61487524fd89780001f1528f
https://vocus.cc/article/61487580fd89780001f154d7
https://vocus.cc/article/614875acfd89780001f155f5
https://vocus.cc/article/6148771efd89780001f15e8f
https://vocus.cc/article/6148769bfd89780001f15b98
https://vocus.cc/article/6148765afd89780001f159ff
https://steemkr.com/movies/@semangateh/watch-hd-full-movie-online-for-free
https://ecency.com/movies/@freehd/watch-hd-full-movie-online
https://paiza.io/projects/dVO1BbEpdiUkHdEsmhQoAA?language=php
https://onlinegdb.com/5OMiTmxoO
https://jsfiddle.net/simbah/k0bupqn4/1/
https://www.peeranswer.com/question/61487bce434d6228725f65e4
https://ideone.com/bhFUiU
http://paste.jp/e377ed3d/
https://rentry.co/oo378
https://paste.centos.org/view/98d9e7c8

- This book completely transformed the way I work and would never have been able to finish a book, let alone a myriad of other things without cultivating

- Home-school would be the expression supplied for the instruction and learning of a child in the possess dwelling. This can be normally

- Season 3 of Selling Sunset dropped on Netflix on August 7, a few short months after Season 2 started streaming. But due to the coronavirus pandemic, many aaa

- Biden administration has continued expelling adults who try to cross the border under a coronavirus-related public health declaration enacted